 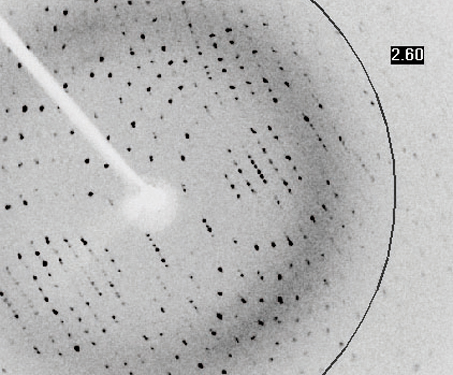
After each blast of X-rays, lasting from a few seconds to several hours, the researchers precisely rotate the crystal by entering its desired orientation into the computer that controls the X-ray apparatus. The electronic detector is the same type used to capture images in a digital camera. The crystal scatters the X-rays onto an electronic detector. Crystallographers aim high-powered X-rays at a tiny crystal containing trillions of identical molecules. X-ray crystallography is used to investigate molecular structures through the growth of solid crystals of the molecules they study. The three components in an X-ray crystallographic analysis are a protein crystal, a source of x-rays, and a detector. Our results define the structure of mammalian Pol II in its functional state, indicate that previous crystallographic analysis of yeast Pol II is relevant for understanding gene transcription in all eukaryotes, and provide a starting point for a mechanistic analysis of human transcription.X-ray crystallography can reveal the detailed three-dimensional structures of thousands of proteins. 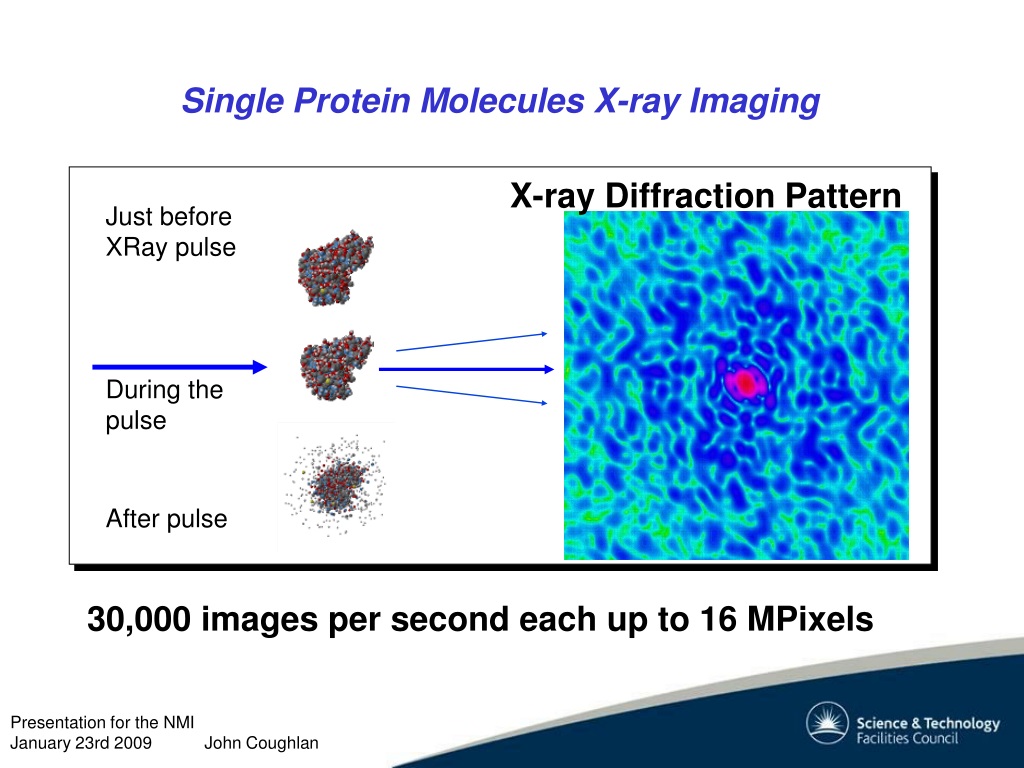
This position of upstream DNA allows for binding of the general transcription elongation factor DSIF (SPT4-SPT5) that we localize over the active centre cleft in a conserved position on the clamp domain of Pol II. Upstream DNA emanates from the active centre cleft at an angle of approximately 105° with respect to downstream DNA. DNA downstream of the transcription bubble contacts a conserved 'TPSA motif' in the jaw domain of the Pol II subunit RPB5, an interaction that is apparently already established during transcription initiation.sup.7. Binding of nucleic acids to the polymerase involves 'induced fit' of the mobile Pol II clamp and active centre region. The obtained atomic model closely resembles its yeast counterpart, but also reveals unknown features. We use bovine Pol II, which is identical to the human enzyme except for seven amino-acid residues. Here we report the 3.4 Å resolution cryo-electron microscopy structure of mammalian Pol II in the form of a transcribing complex comprising DNA template and RNA transcript. Structural studies of mammalian Pol II, however, remain limited to low-resolution electron microscopy analysis of human Pol II and its complexes with various proteins.sup.4,5,6,7,8,9,10. The Pol II structure is known at high resolution from X-ray crystallography for two yeast species.sup.1,2,3. RNA polymerase (Pol) II produces messenger RNA during transcription of protein-coding genes in all eukaryotic cells. The overall structure resembles previously determined crystal structures of the yeast Pol II elongation complex, but also reveals a number of differences in domain and substrate orientations that will provide a starting point for mechanistic understanding of human transcription. Here Patrick Cramer and colleagues use cryo-electron microscopy to determine the first high-resolution structure of mammalian (bovine) Pol II in the form of an elongation complex with a bound DNA template and RNA transcript. The high-resolution structure of yeast Pol II is known, but structural investigation of the mammalian enzyme have so far been limited to low-resolution studies. 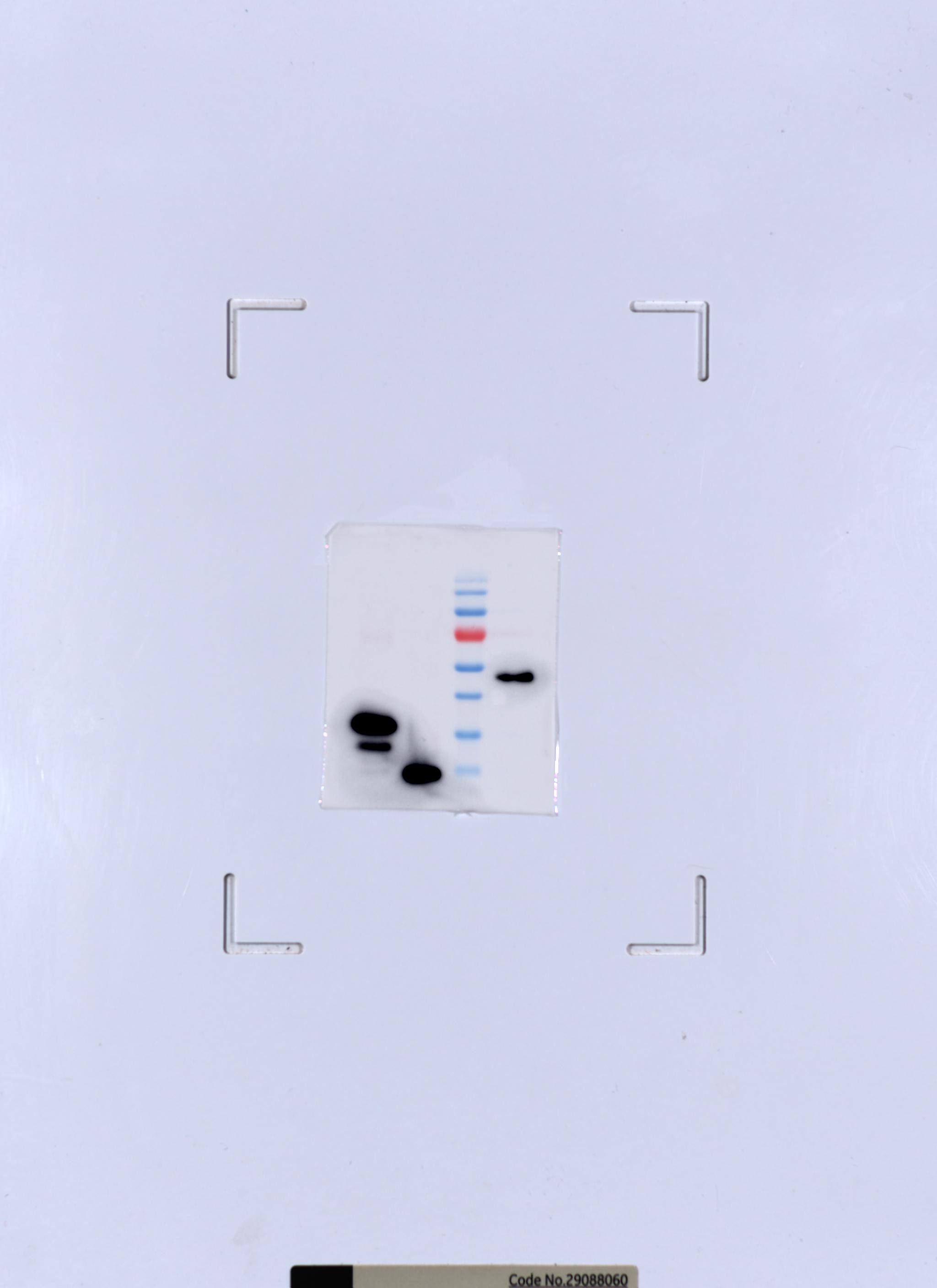
Mammalian RNA polymerase II structure Eukaryotic RNA polymerase II (Pol II) produces mRNA during transcription of protein-coding genes. Abstract : The first high-resolution, cryo-electron microscopy structure of mammalian RNA polymerase II, in the form of a transcribing complex comprising DNA template and RNA transcript. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
